|
The vast majority of the known elements are metals. 
Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. /accurate-illustration-of-the-periodic-table-82020791-57cc76f23df78c71b66efbd7.jpg)
The distinction between metals and nonmetals is one of the most fundamental in chemistry. Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. \) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line). The ionic radius values are shown in the chart below. The increasing number of protons increases the attractive electrostatic force, resulting in smaller radii. The ionic radius decreases as the number of protons increases. The ions are arranged according to their increasing number of protons. The ionic radius values of isoelectronic ions N 3-, O 2-, F –, Na +, Mg 2+, and Al 3+ are given in the table below for 6-coordinate, octahedral geometry. An increase in protons increases the electrostatic force and brings the electrons closer to the nucleus.Īlso, from Al 3+ to P 3-, there is a sudden jump in the ionic radius due to an extra shell of electrons. The cations P 3-, S 2-, and Cl – are isoelectronic, and their ionic radius decreases from left to right. The electrons are held closer to the nucleus, and the radius shrinks. The reason is that an increase in the number of protons leads to an increased attractive electrostatic force between the nucleus and the valence electrons. Their ionic radii decrease from left to right. Ions having the same electron configuration are isoelectronic. The cations Na +, Mg 2+, and Al 3+ have the same electron configuration. Three cations and three anions are listed together with their number of protons, electron configuration, and ionic radius for 6-coordinate, octahedral geometry. Hence, this trend must be studied separately for cations and anions.Ĭonsider the period 3 elements which are shown in the table below. Horizontal: Ionic Radius Trend Across a Periodĭue to an atom’s unique ability to gain or lose electrons, the ionic radius trend across a period is not precisely similar to the atomic radius trend. The following table gives the ionic radius values of group 17 elements for 6-coordinate, octahedral geometry. The following table gives the ionic radius values of group 1 elements for 6-coordinate, octahedral geometry. 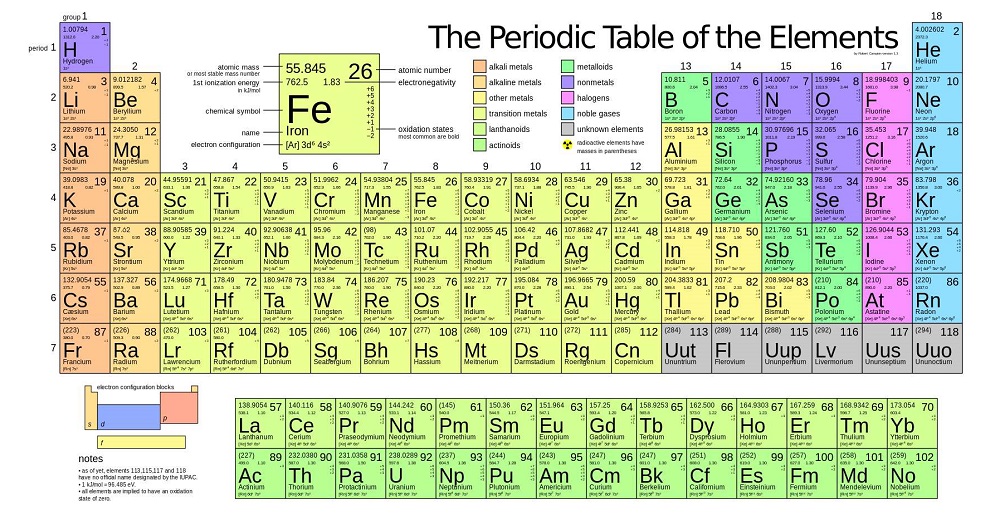
Thus, the ionic radius has the same vertical trend as the atomic radius. 
As a result, the attractive electrostatic force between the nucleus and the valence electrons also decreases, and the electrons remain further away. An effect of shielding is that the valence electrons now see a nucleus that is reduced in charge, known as the effective nuclear charge (Z eff). The inner electrons shield the outer electrons, and the shielding increases down the group. The reason is that when an extra shell of electrons is added to the atom, the valence electrons get further away from the nucleus. Vertical: Ionic Radius Trend Down a Groupĭown a group, the atomic size and the ionic radius increases for ions with the same charge, i.e., same oxidation state. This pattern is known as the ionic radius trend. The elements display a specific pattern in their ionic radius across a period and down a group in the periodic table. Their ionic radius trend is discussed below. Most periodic table elements, except noble gases, lose or gain electrons to form ions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
